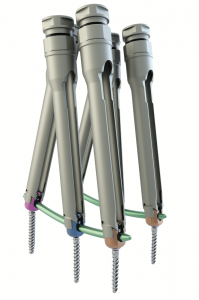
mont blanc MIS
The mont blanc MIS implants consist of various forms and sizes of screws, locking screws and rods.
The mont blanc MIS implants can be combined for creating a posterior spinal implant system in a minimally invasive approach.
They are used with dedicated instruments which allow preparation of the site, insertion of implants and removal.
The mont blanc MIS implants are supplied for single use.
Implants material: titanium alloy (ISO 5832-3).
Instruments material: stainless steel, silicone, polyphenyl sulfone, POM, nitinol, ABS and polypropylene.
The mont blanc MIS system is intended to provide immobilization and stabilization of spinal segments in skeletally mature patients as an adjunct to fusion in the treatment of acute and chronic instabilities or deformities of the thoracic, lumbar and sacral spine.
This mont blanc MIS system is intended for posterior noncervical pedicle fixation and non-pedicle fixation (from T1 to S1) utilizing a percutaneous minimally invasive approach for the following indications in skeletally mature patients:
- degenerative disc disease (defined as back pain of discogenic origin with degeneration of the disc confirmed by history and radiographic studies);
- spondylolisthesis;
- trauma (i.e., fracture or dislocation);
- spinal stenosis;
- deformities (i.e., scoliosis, kyphosis, and/or lordosis);
- spinal tumor,
- pseudoarthrosis; and
- failed previous fusion.
The mont blanc MIS system is intended to be used in a posterior minimally invasive approach.
The mont blanc MIS system is intended to be used with autograft and/or allograft.
When used in adolescent patients, the mont blanc MIS system is indicated as an adjunct to fusion to treat idiopathic scoliosis.